You will be seen soon after the procedure to remove the contact lens and you will remain on eye drops for a few weeks. You will be seen at 1 month, 3 months, 6 months and one year. Following this it is important that you be seen on an annual basis by your optometrist as you normally would.
Collagen Crosslinking with Riboflavin
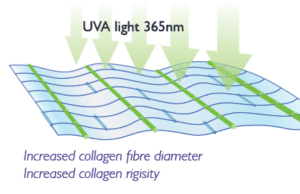
Ultraviolet light is used to promote increased cross-linking between collagen fibres within the cornea.



What is the CXL procedure?
Ultraviolet light is used to promote increased cross-linking between collagen fibres within the cornea. Increasing the amount of cross-linking results in increased strength or rigidity of the cornea.

Keratoconus and corneal ectasia are conditions where the cornea bulges and becomes “cone” shaped. This results in astigmatism which can become irregular and impair vision even with glasses. Often contact lenses are the only option available to provide improved vision. Should patients become contact lens intolerant because of disease progression, then surgical options may need to be considered. These options include implantation of Intacs or Ferrara intracorneal rings or partial or full thickness corneal transplants. Strengthening the cornea by cross-linking its building blocks (collagen) can arrest progression of keratoconus and has also been reported to partially reverse the corneal steepening that has already taken place.
What does the procedure involve?
The cornea which is the front transparent structure of the eye is first anaesthetised using drops. The procedure is performed in the clinic in a semi-reclined chair or stretcher. Strong antibiotics are also used to prevent infection. The mucous surface of the cornea (epithelium) is disrupted using a special instrument (Daya Epithelial Disruptor) and then Riboflavin (Vitamin B2) drops are placed in the eye every few minutes.

How long does it take for the procedure to work?
Cross-linking takes place as a result of exposure to Ultraviolet light. The cornea increases in rigidity soon after the procedure although the process of crosslinking continues on for a period of a few days afterwards. The effect on corneal shape takes longer but flattening does not occur in all eyes that have had treatment. A satisfactory result will be arresting the progress of keratoconus.
FAQs
You may return to wearing lenses after one month. Your lenses may need to be changed if your cornea changes shape.
As the mucous lining of the cornea is disrupted, there is a very small risk of infection. This is rare and prevented through the use of antibiotics before and after the procedure.
“Ultraviolet: A light used in this procedure is not harmful to the eye in measured doses”
Ultraviolet: A light used in this procedure is not harmful to the eye in measured doses. Ultraviolet C light (in sunlight) is potentially harmful. The Light emitting diodes used in the 3C-R device is of a wavelength that is not harmful. Furthermore, light emission is carefully measured and calibrated prior to each treatment. There is also a self diagnostic check on the device which prevents use in case of a malfunction. There have been concerns about toxicity to the retina (at the back of the eye) however the Riboflavin pigment in the cornea and front of the eye, absorbs the Ultraviolet light and in effect stops the light from being transmitted to the retina.
Our Consultants
Author Information
Authored by Sheraz Daya MD FACP FACS FRCS(Ed) FRCOphth, Consultant Ophthalmic Surgeon & Medical Director, June 2019.
Next review due June 2025.